- Home
- Services
- About
- News
- Contact
- What is another word for issue or problem
- Neue haas unica adobe
- Wolfquest download for windows 7
- Pc emulator for mac
- What products at relux in nucleophilic substitution
- How to create an electronic signature in preview
- Best art tablets 2017
- Deool marathi movie download hd
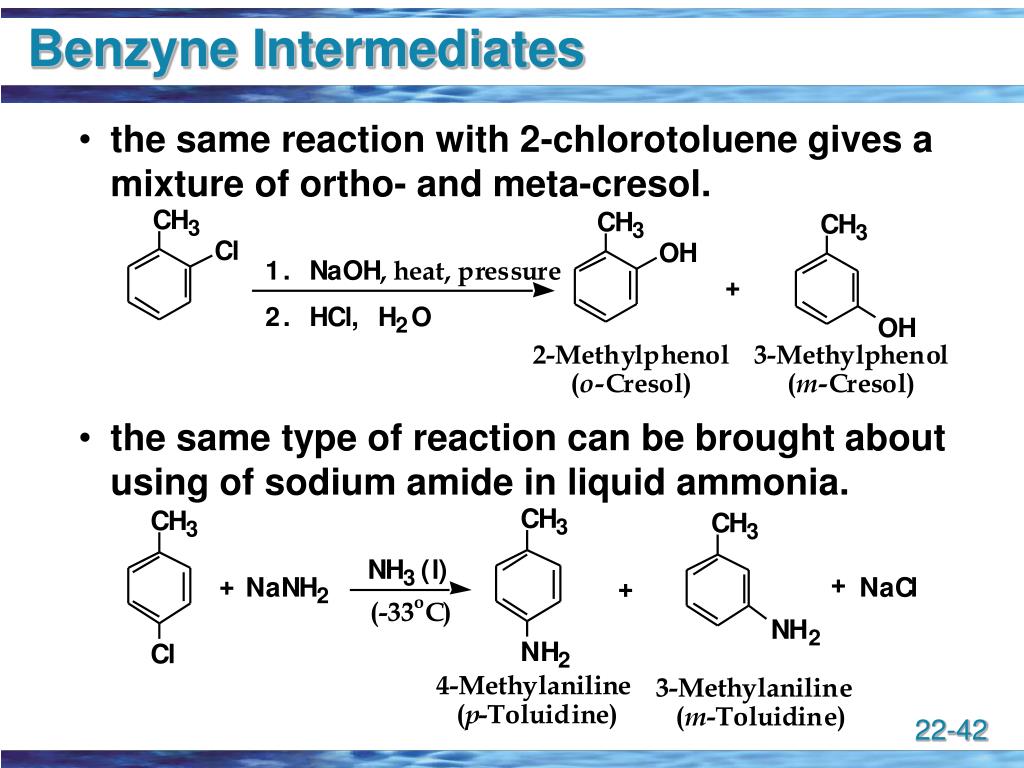
What is the reason for this change of reactivity and, in general, what is the mechanism of nucleophilic aromatic substitution? We learn that the carbon-fluorine bond is the strongest and the iodide being polarizable was the best leaving group. So, the more electronegative the halogen, the better leaving group it is in a nucleophilic aromatic substitution.Īnd this is the opposite of what we learned in the S N1 and S N2 reactions. We are going to talk about the details of the mechanism below, but for now, let’s also mention that the reactivities of aryl halides increases, depending on the leaving group, in the following order: This is the only orientation supporting the resonance stabilization of the negative charge in the transition state. One requirement for these reactions is the ortho or para position of the electron-withdrawing group to the leaving group. The nitro group is typically used as the electron-withdrawing group even though other resonance-withdrawing groups such as carbonyls can also activate the ring toward a nucleophilic attack.Īs for the nucleophile, a variety of charged and neutral strong nucleophiles such as –OH, –OR, –NH 2, –SR, NH 3, and other amines can be used.

X here is the leaving group and the EWG stands for electron-withdrawing group which is there to activate the ring by making it electron-deficient. However, some aryl halides with a strong electron-withdrawing substituent(s) on the ring can undergo nucleophilic substitution ( S N Ar) instead of electrophilic substitution:
We have seen that most reactions of aromatic compounds involve electrophilic substitutions because the π electrons make the aromatic ring electron-rich and therefore, nucleophilic.
- Home
- Services
- About
- News
- Contact
- What is another word for issue or problem
- Neue haas unica adobe
- Wolfquest download for windows 7
- Pc emulator for mac
- What products at relux in nucleophilic substitution
- How to create an electronic signature in preview
- Best art tablets 2017
- Deool marathi movie download hd
